In the realm of atomic structure and periodic table elements, the stability and chemical inertness of noble gases often captivate both students and seasoned chemists alike. Neon, as a prominent member of this group, exemplifies the qualities that arise from its electron configuration. Yet, amidst advancements in quantum chemistry and spectroscopic analysis, questions persist—particularly, "How many valence electrons does neon have today?" This inquiry, seemingly straightforward, opens a window into the nuanced understanding of atomic valence, electronic behavior, and their implications for both fundamental science and practical applications.
Understanding Neon’s Electronic Configuration and Its Valence Electron Count

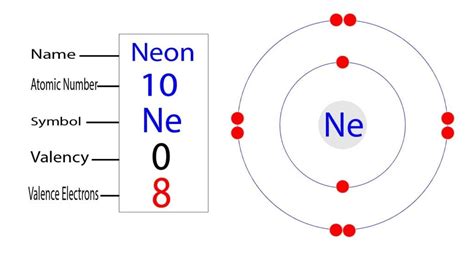
Neon, with an atomic number of 10, belongs to Group 18 (the noble gases) in the periodic table. Its electronic configuration in the ground state is 1s2 2s2 2p6. This configuration indicates that the first energy shell (n=1) is fully occupied by two electrons, while the second shell (n=2) contains eight electrons, filling the 2s and 2p orbitals completely. Consequently, neon’s outermost shell—the second shell—is saturated with eight electrons, fulfilling the octet rule.
Electron Shells, Valence, and Atomic Stability
The concept of valence electrons traditionally refers to the electrons in the outermost electron shell of an atom that participate in chemical bonding. For neon, the second shell acts as this valence shell, and given its full occupancy of eight electrons, neon is chemically inert. This inertness stems from the closed-shell configuration, rendering neon largely unreactive under standard conditions.
| Category | Data and Context |
|---|---|
| Atomic number | 10 |
| Electrons in the first shell | 2 |
| Electrons in the second shell | 8 |
| Valence electrons (traditional definition) | 8 |

Contemporary Perspectives and Quantum Mechanical Insights into Neon’s Valence

While classical models accurately assign neon’s valence electrons as eight occupying the 2p orbital, quantum mechanical frameworks enrich this picture. The electron cloud of neon embodies a probabilistic distribution governed by wavefunctions, with electrons occupying orbitals characterized by specific quantum numbers. Advances in quantum chemistry computational methods, including density functional theory (DFT), reaffirm that the effective valence—the electrons most involved in interactions—is still represented by the electrons in the second shell, totaling eight.
Valence in Excited States and Ionization Dynamics
It’s noteworthy that in excited states, the electron configuration of neon can transiently deviate, such as upon promotion of an electron from the 2p to higher orbitals. Similarly, ionization experiments demonstrate that removing electrons typically affects the outermost electrons first. In these contexts, the count of valence electrons can fluctuate momentarily, but in the stable ground state, the total remains firmly at eight.
| Application Area | Insight |
|---|---|
| Spectroscopy | Valence electron count influences spectral lines, with neon’s characteristic emission lines arising from electronic transitions involving outer-shell electrons. |
| Chemical Reactivity | Neon's inertness is directly linked to its full valence shell, confirming that its valence electrons are indeed the eight in the second shell. |
| Material Science | Neon’s stable valence electron configuration underpins its use in lighting, laser technology, and as a noble gas buffer in cryogenics. |
Implications of Valence Electron Count for Practical and Scientific Applications
Understanding that neon possesses eight valence electrons today is more than a theoretical curiosity; it underpins its applications across multiple fields. Neon’s chemical inertness, rooted in its full valence shell, makes it ideal for use in neon lighting, where electron transitions produce vibrant colors without reactivity complications. Similarly, in high-precision scientific environments, this electron configuration ensures predictable behavior under electromagnetic influences, facilitating reliable measurements and experimental manipulations.
Historical Evolution of Electron Configuration Knowledge
The initial discovery of neon in 1898 by Morris W. W. Ramsay and William Ramsay, through spectroscopic analysis, cemented the understanding of its atomic composition. Over the decades, advances in quantum mechanics and computational chemistry have refined this knowledge, with modern spectroscopic techniques providing direct evidence of the electron counts predicted by early models. Despite these technological revolutions, the fundamental count of eight valence electrons for neon remains unaltered, illustrating the robustness of foundational atomic theory.
| Historical Milestone | Impact |
|---|---|
| 1898 - Discovery of Neon | Established initial spectral understanding, linking observed lines to electron transitions. |
| 1930s - Quantum Mechanical Models | Allowed precise calculation of electron configurations and the concept of noble gas invariance. |
| Recent - Spectroscopic verification | Confirmed the electron counts and electronic structure models with high resolution. |
Current Challenges and Future Directions in Atomic and Molecular Analysis
Despite the established understanding, ongoing research explores subtler effects—like polarization, relativistic effects in high-energy environments, and interactions with external electromagnetic fields—that may slightly influence the effective valence electron behavior. Emerging techniques such as ultrafast spectroscopy and quantum simulations aim to capture these transient phenomena, potentially refining our conception of what constitutes ‘valence electrons’ in exotic states or extreme conditions.
Advances in Computational Quantum Chemistry
Next-generation algorithms now incorporate electron correlation effects at unprecedented levels, enabling the simulation of reactive processes involving neon with fine detail. These tools support the understanding that, even under dynamic excitation or in plasma states, the core count of eight valence electrons remains a reliable reference point.
Conclusion: The Immutable Number in a Dynamic Context

Today, as ever, neon’s number of valence electrons holds steady at eight, anchoring our comprehension of its chemical inertness and spectral properties. This constancy exemplifies the enduring strength of foundational atomic principles, even amidst the rapid evolution of experimental techniques and theoretical models. For scientists, educators, and industry professionals alike, the reaffirmation of neon’s valence electron count underscores a broader lesson: while our methods and applications evolve, some truths—like neon’s steadfast eight valence electrons—persist unchallenged, guiding innovation with their unwavering clarity.
How do we verify the number of valence electrons in neon today?
+Using advanced spectroscopic techniques such as X-ray photoelectron spectroscopy and quantum chemical calculations, scientists can directly observe and confirm the electron configuration, reaffirming the count of eight valence electrons in neon’s outer shell.
Has the understanding of neon’s valence electrons changed over time?
+No, the core concept of neon having eight valence electrons has remained consistent. Technological and theoretical advancements have refined our understanding, but the fundamental count remains unchanged, thanks to the stability of its electronic configuration.
Why is the valence electron count important in practical applications?
+Valence electrons determine an element’s chemical reactivity, spectral properties, and bonding behavior. Neon’s full valence shell makes it inert, influencing its use in lighting, lasers, and cryogenic environments where stability and predictable behavior are essential.